November 11, 2021, by bmillar
Accelerated TMS for Pain Inhibition: The BoostCPM Study
What’s this & why would it help with pain?
There is more and more evidence that effective treatments need to focus on any disturbed communication between brain areas that are fundamental to the perception of pain. Logically, if abnormal brain connectivity patterns are reversed, this should help to alleviate chronic pain.
Transcranial Magnetic Stimulation (TMS) offers the possibility to do so non-invasively, safely and with practically no side-effects. It has already been approved by the National Institute of Clinical Excellence (NICE) for treatment-resistant depression.
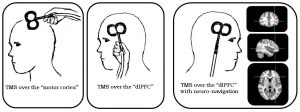
Basically, during an intervention session a coil is placed next to the head to send magnetic pulses at a specific spot in the brain to stimulate some nerve cells to change their behaviour. That’s why it’s called neuromodulation.
Hair not displayed for better illustration. Hair, skin, and skull are not affected by this in any way.
Many studies recently have found this technology to be promising results for pain relief. Our group at the University of Nottingham aims to improve the success rate with an accelerated TMS protocol.
![]()
Note that some participants in this study will receive inactive (placebo) and some will receive active TMS treatment. Participants will not be told before the end of the study whether they received active or placebo TMS intervention.
What would you need to do if you take part?
- Take part in an initial telephone consultation
- Complete questionnaires
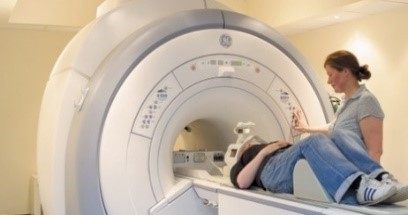
- Have an MRI scan of your brain (see info below)
- Undergo TMS application
- Undergo sensory testing. This will be done without any harm to your skin and can be stopped instantly, should you not like to tolerate it. You will get more info before participating.
- Participants are also asked to complete online pain and wellbeing questions up to 16 weeks.
- As the study aims to assess the benefits of a TMS stimulation protocol for longer term effects, the core study schedule is fairly intense as the accelerated protocol substitutes a month’s worth of daily TMS.
- There are 6 visits in total: 2h for the first visit, 4 TMS intervention days with 5h each but with 4x 50 minutes breaks, 2h for the final Refreshments and comfy furniture will be provided in the breaks where you can read or entertain yourself as you like.
What’s MRI?
MRI, short for Magnetic Resonance Imaging, has been used in hospitals and research settings for about 25 years and is a safe technique, which produces pictures without ionizing radiation (in contrast to x-rays). This is how a scanner looks & an example brain image:
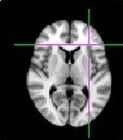
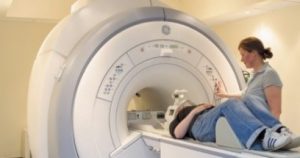
MRI does not cause any side-effects. Still, you will always be able to tell us to stop, should you dislike it.
Who are we looking for?
People who
- Have moderate-severe chronic knee pain
- Have no pacemaker or artificial heart valve).
- Are not anxious in tight spaces (MRI is a tube)
- Do not have epilepsy, seizures, convulsions or family history thereof
- Have time to participate in an intense study
Where does the study take place?
- QMC Hospital, Nottingham, NG7 2UH.
- We can’t pay for participation but refreshments will be provided for free, and travel & parking expenses will be reimbursed.
- In some cases we can also pay for taxi fares.
Who funds this study? Versus Arthritis UK (charity)
Who are we?
We are part of Nottingham University’s Pain Centre which has been funded by Versus Arthritis for the past 10 years.
If you are curious about the Pain Centre’s research, feel free to browse https://www.nottingham.ac.uk/paincentre/
To get more info on this particular study and get your questions answered, ask the study team:
Email: msk-recruitment@nottingham.ac.uk
Telephone: 0115 8231676
No comments yet, fill out a comment to be the first

Leave a Reply