
September 11, 2017, by Emma Rayner
Celebrating 20 years of improving treatment of skin diseases
“It’s like putting an octopus into a string bag,” says Professor Hywel Williams, the visionary founder of Cochrane Skin, a global network of people committed to producing reviews of trials relating to skin conditions, which celebrates its 20th anniversary this month. “The octopus represents the many arms of global dermatology research but gathering it all together, assessing it and extrapolating the truth to improve patient treatment is another challenge altogether.”
Professor Williams is one of the Pied Pipers of the skin world who, through imagination, passion and drive for progress in his field, and supported by a fantastic team, has led Cochrane Skin through two decades of gathering and summarising the best evidence on the prevention and treatment of skin diseases from across the world, in order to help clinicians and patients make informed choices about treatment.
The group is part of the wider Cochrane network of groups who organise systematic reviews of primary research that covers all areas of health and health policy. Cochrane is internationally recognised as setting the highest standards in evidence-based health care resources. Cochrane Skin is grant-funded by the National Insitute for Health Research (NIHR) Systematic Reviews Programme.
Inspired by a call to action from one of the original Cochrane founders, Sir Iain Chalmers, in the British Medical Journal in 1992, Professor Williams set about building Cochrane Skin, which was officially registered with Cochrane (originally The Cochrane Collaboration) on 12th September 1997.
The editorial base of Cochrane Skin resides within the Centre of Evidence Based Dermatology (CEBD) at the University of Nottingham, which acts as an essential “cog” in summarising uncertainties when sufficient data is available, or more often that not, highlighting key gaps in clinical research. These gaps can then be picked up by the UK Dermatology Clinical Trials Network cog at the CEBD, such as the recently completed NIHR study of doxycycline antibiotics for the treatment of pemphigoid that was initially highlighted in a Cochrane Review.
At present more than 1,300 people from 61 different countries contribute to the work of the Cochrane group, with an established Satellite group in France and others planned in South America and the USA.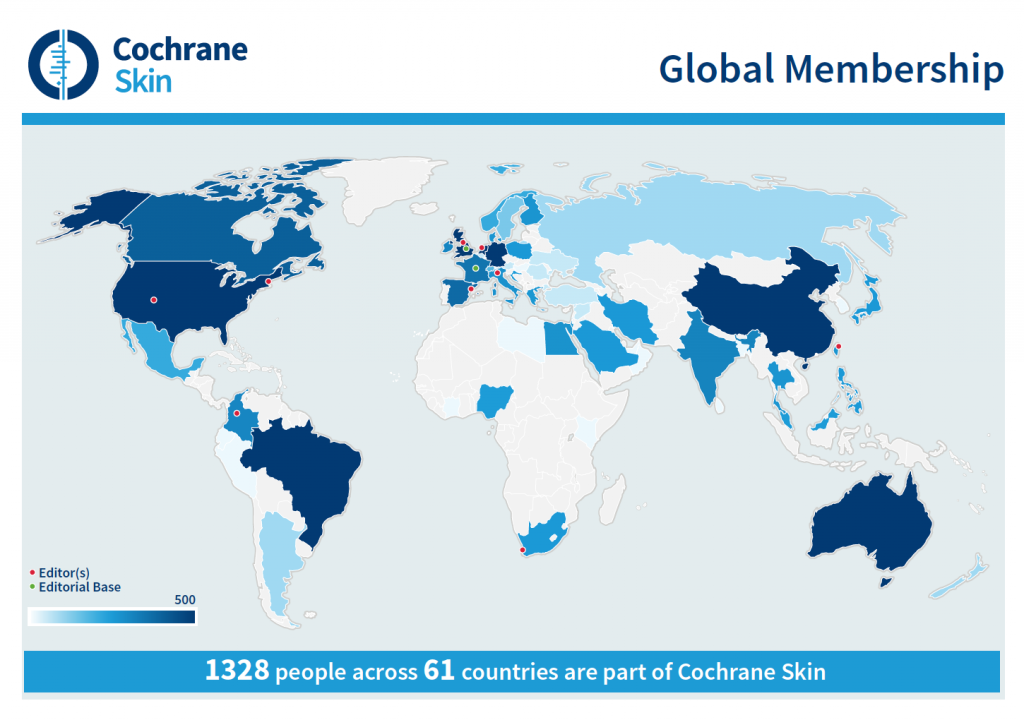
“By systematically reviewing and summarising all relevant clinical trials, Cochrane Skin tries to drive up the quality of primary research into skin disease prevention and treatments,” said Professor Williams. “A lot of dermatology clinical trials have addressed unimportant questions that mean little to patients, and which have been designed and reported badly. By sorting out the wheat from the chaff, Cochrane Skin draws a line in the sand in order to stimulate better primary research.
“Evidence-based dermatology has come a long way in the last 20 years, and Cochrane Skin has played a very important part in informing guidelines, stimulating new clinical trials, undertaking methodological research, encouraging better reporting of trials in journals as well as capacity building and knowledge mobilisation. Patients (or ‘consumers’) have always been at the heart of Cochrane Skin from its outset, and nearly all Cochrane Skin reviews include a patient co-author. Together, we have made invaluable contributions to the evidence base for skin disease management.”
The effects of the 120 plus published Cochrane Skin Reviews have been far-reaching and have had real impacts on patient care. These include skin cancer, skin allergies and acne, blistering diseases, hair disorders like alopecia, fungal infections and psoriasis as well as tropical parasitic diseases. In addition the scope of Cochrane Skin includes areas that may be perceived as cosmetic, such as skin ageing.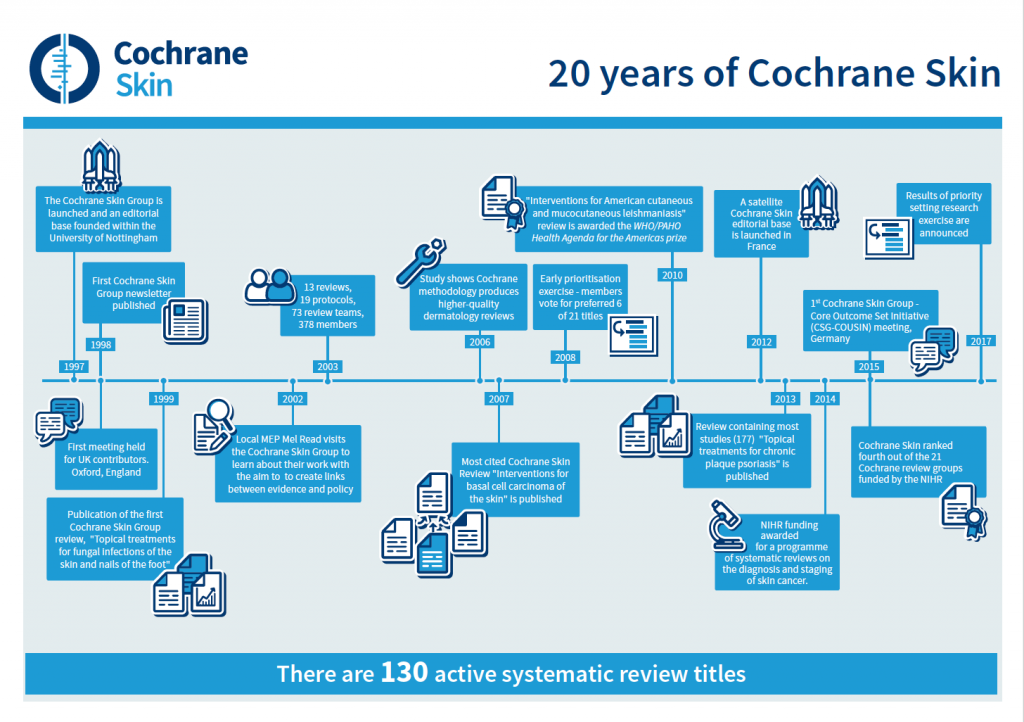
In 2014, Cochrane Skin and the Biostatistics, Evidence Synthesis and Test Evaluation team in Birmingham was awarded NIHR funding for a programme of systematic reviews on the diagnosis and staging of skin cancer, i.e. melanoma and keratinocyte skin cancers (basal cell and cutaneous squamous cell carcinomas, also termed ‘non-melanoma’ skin cancers). Some overviews will also be created to inform policy, such as the National Institute for Health and Care Excellence (NICE) Guidelines. The project is being led by Dr Jac Dinnes from the Birmingham team and is scheduled to complete by the end of 2017. Such diagnostic test accuracy reviews are just one example of more methodologically challenging systematic reviews, which Cochrane Skin hopes to produce more of going forward. Reviews including techniques such as network meta-analysis and individual patient meta-analysis are also under development as Cochrane enters a new era of working across larger networks of review groups.
 From the outset, Cochrane Skin has closely involved patient groups in its work. Maxine Whitton MBE who is a Co-Founder of The Vitiligo Society, was formerly Chairman and is now a Patron, has had the skin pigmentation condition, vitiligo since childhood. Maxine said:
From the outset, Cochrane Skin has closely involved patient groups in its work. Maxine Whitton MBE who is a Co-Founder of The Vitiligo Society, was formerly Chairman and is now a Patron, has had the skin pigmentation condition, vitiligo since childhood. Maxine said:
“Professor Hywel Williams encouraged and supported me in producing the first review of interventions for vitiligo. This review retrieved just 19 randomised trials, mostly of poor quality, published since 1966, highlighting the need for more research and well-designed trials as the basis for informed decisions on treatments. Under the auspices of Cochrane Skin we conducted two further reviews published in 2010 (57 trials) and 2015 (96 trials). The increased number of randomised trials assessed for the review reflected the increase in research and also in the number of different treatment options now being used for vitiligo, although sadly the majority of the studies were still badly designed.
“The review has stimulated further vitiligo research and projects in which I and members of the Society have been involved, such as the development of core outcome sets and patient related outcome measures and a Priority Setting Partnership conducted by the James Lind Alliance. The Vitiligo Society has also helped in the recruitment of participants for a pilot study of psychological interventions for vitiligo and also a large multi-centre trial combining topical treatment and NB-UVB for vitiligo (Hi-Light) based at Nottingham University, which is ongoing.”
Coinciding with Cochrane Skin’s anniversary is the culmination of a large-scale prioritisation exercise, which the group has undertaken in the last few months to determine the reviews or updates that should be concentrated on over the next three years. A wide range of stakeholders were consulted, with a view to identifying which titles were either key to informing guidelines or policy, relevant to skin conditions in low- to middle-income countries, or of maximum clinical importance and greatest relevance to patients and health care funders. This exercise is the result of an ever-expanding portfolio and the pressure that puts on the limited capacity of the editorial base to manage the review process and ensure quality and relevance.
Whether you are a healthcare professional or a patient or a member of the public, there are many ways in which people can become involved in Cochrane – for example, as volunteer searchers through Cochrane Crowd, or by refereeing new Cochrane protocols and reviews, as well as authoring a review that has been prioritised by the relevant group. Information on how to join the group is available at http://join.cochrane.org/.
Cochrane Skin will be celebrating its 20th anniversary as the first Global Evidence Summit (GES) takes place in Cape Town, South Africa (13th – 16th September 2017). This event sees Cochrane join with four other leading organisations – the Guidelines International Network, The Campbell Collaboration, the International Society for Evidence-based Health Care, and the Joanna Briggs Institute – to focus on the opportunities and challenges facing low- and middle-income countries. The theme of the GES, ‘Using Evidence. Improving Lives’, will highlight and promote evidence-informed approaches to health policy and development, offering the most cost-effective interventions.
No comments yet, fill out a comment to be the first


Leave a Reply