February 26, 2021, by School of Medicine
50 at 50: No research about me without me! Welcoming the patient and public voice to research
A lot has happened in healthcare and research over the last 50 years since the University of Nottingham Medical School was born. Not just in breakthrough treatments and diagnostics, blockbuster drug development and in tackling our first pandemic since 1918; but also in opening up medicine and healthcare to include the views and experiences of patients and the public.
For years, the doctor’s word was final: a paternalistic approach where healthcare was ‘done’ to patients. A similar approach was taken in research. Researchers ‘knew’ what patients needed. They ‘knew’ what questions needed answering. While this may often have been done with good intentions, important perspectives and questions were not being raised or addressed.
Researchers often do not have the health condition or disease that they study. They don’t deal with the day-to-day impacts, nor navigate the healthcare or social care system to access treatment or support. While researchers may be expert in research methods and the published literature, they lack the insight that comes from experiencing the condition and using services as a patient or carer.
Patient experts have expertise through relevant lived experience of the health condition under study. Members of the public hold broader perspectives of healthcare and can share their impressions of research as someone from the ‘outside’ looking in. Patients and public play a valuable role in ensuring that research is focused on what matters to patients and is communicated effectively to a wide range of audiences. This is not as participants in a ‘fact-finding’ exercise but as equal partners in a team designing, running and communicating research.
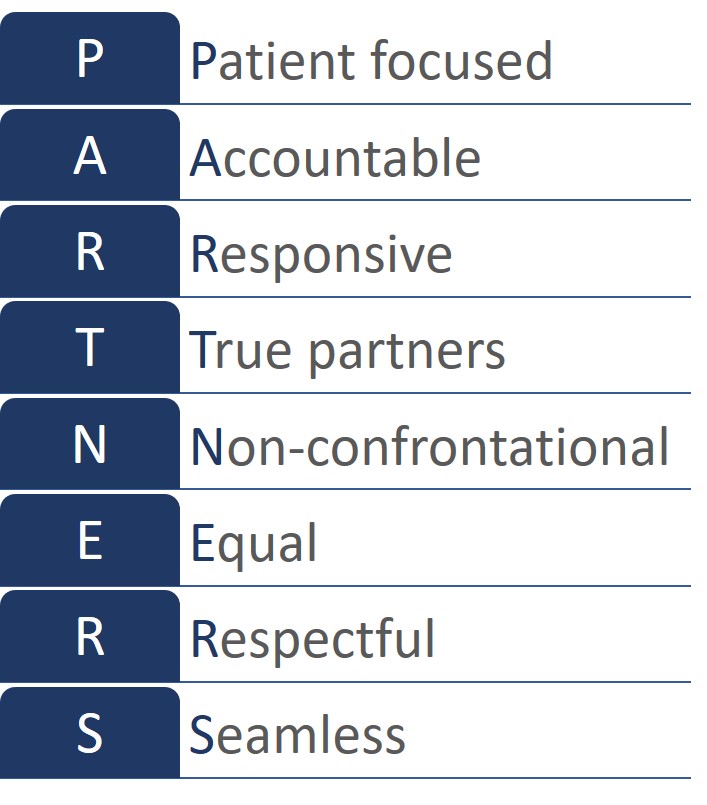
The Nottingham Stroke Research Partnership Group’s philosophy is summarised in the acronym PARTNERS.
Today, involving patients and the public in research is increasingly seen as the ‘norm’ and many funders, such as the National Institute for Health Research (NIHR) and medical charities, expect to see patient and public involvement, or PPI, included in study plans.
Researchers in the School of Medicine work with patients and the public who volunteer their time and expertise to studies. Some examples are below.
The Nottingham Stroke Research Partnership Group (@astroke or NSRPG) in the Division of Rehabilitation, Ageing and Wellbeing, is a group of stroke survivors and carers who advise Nottingham stroke researchers on study ideas and plans and take an active role in running the research. Formed in 2004, the NSRPG have patient and academic co-chairs, exemplifying the group’s equal partnership philosophy.
 The Centre of Evidence Based Dermatology (CEBD) has a long-standing history in involving patients and carers in research. A patient panel was established in 2009 and now has more than 30 members across the UK, who live with a range of skin conditions. Members contribute to research in a wide variety of ways.
The Centre of Evidence Based Dermatology (CEBD) has a long-standing history in involving patients and carers in research. A patient panel was established in 2009 and now has more than 30 members across the UK, who live with a range of skin conditions. Members contribute to research in a wide variety of ways.

Hearing research project meeting with public research partners (left to right) Veronica Colley, Claire Ward and Julia Brown
Hearing sciences, in partnership with the Nottingham Biomedical Research Centre, involve people with hearing difficulties as equal partners. Founded in 2015, our patient and public steering group agree strategy and oversee an internal funding scheme. They join Hearing’s Research Advisory Group, advising researchers alongside other experts. ‘Ear Facts’ webpages also drew upon members contributions. Researchers led James Lind Alliance Priority Setting Partnerships with the public on tinnitus, mild to moderate hearing loss and hyperacusis.
The Musculoskeletal Patient Advisory Group offers members the opportunity to be actively involved in various musculoskeletal research roles, co-producing studies in the musculoskeletal theme of the Nottingham Biomedical Research Centre, Pain Centre Versus Arthritis and the Centre for Sport, Exercise and Osteoarthritis Research Versus Arthritis. Researchers and potential Patient Advisory Group members can email MS-msk-ppi@nottingham.ac.uk for more information.
Patient and public involvement is still a relatively novel concept to some and so the School patient and public involvement working group, led by Dr Rebecca Fisher, with Dr Adele Horobin and Dr Manjeet Mundey, have focussed on education and training.
 Education and training is also the focus of the East Midlands Sharebank, which was formally established in 2018. Chaired by Dr Adele Horobin, it brings together staff and public representatives from research organisations across the East Midlands to network, share ideas, training and resources on patient and public involvement. One timely resource is the Sharebank’s ‘Working with patients and the public in a virtual world’, which is packed with general advice about running online meetings, helpful links, overview of video conference providers and example templates. University staff can find this and related information on their workspace.
Education and training is also the focus of the East Midlands Sharebank, which was formally established in 2018. Chaired by Dr Adele Horobin, it brings together staff and public representatives from research organisations across the East Midlands to network, share ideas, training and resources on patient and public involvement. One timely resource is the Sharebank’s ‘Working with patients and the public in a virtual world’, which is packed with general advice about running online meetings, helpful links, overview of video conference providers and example templates. University staff can find this and related information on their workspace.
Drawing upon the Sharebank network, Rebecca and working group members invite public and staff experts in patient and public involvement to co-produce and co-deliver training to staff and students. This network underpinned the success of our flagship undergraduate module on patient and public involvement in research for medical undergraduate students. The upcoming internal seminar series on patient and public involvement, starting 25 March, will be drawing upon the network’s expertise. See the School of Medicine bulletin for details.
Learning about patient and public involvement is part of many postgraduate courses in the School of Medicine. Public partners Trevor Jones, Michael Prior, Amanda Roberts and Stevie Vanhegan share their perspectives with Dr Joanne Stocks in the Grant Writing Programme led by Dr Paul Leighton for early career researchers. Prof Cathy Merry’s Focus on Fellowships course includes an overview of patient and public involvement for clinical and discovery science researchers, delivered by Dr Manjeet Mundey. While the Nottingham Clinical Trials Unit offers insights for trial staff and researchers through the Fundamentals of Clinical Trials course. The Researcher Academy at the University includes a two-day course on involvement, led by Andy Wragg with Dr Jane Horne and public partners Karen Andrews, Michael Prior, John Shaw and Stevie Vanhegan. Also, the NIHR Research Design Service offers patient and public involvement support, including a funding scheme, to staff and students.
In all, the future is looking bright for the patient and public’s voice in research at the School of Medicine. There is still much to do, but with such examples of good practice, more researchers will be inspired to follow their lead.
Further reading
- Centre of Evidence Based Dermatology blog. Read their most recent post about how they have reviewed their activities in line with the national standards for public involvement in research. Available at: https://www.nottingham.ac.uk/research/groups/cebd/cebd-blog.aspx [Accessed 8 February 2021].
- UK Standards for Public Involvement. Available at: https://sites.google.com/nihr.ac.uk/pi-standards/home [accessed 8 February 2021].
- No research about me without me – why researchers should welcome the patient’s voice. Available at: http://blogs.biomedcentral.com/on-medicine/2018/03/02/no-research-about-me-without-me-researchers-welcome-patients-voice/. This post describes one example of patient and public involvement in hearing research [accessed 8 February 2021].
- University of Nottingham School of Medicine staff intranet workspace for more guidance and resources on patient and public involvement in research. https://workspace.nottingham.ac.uk/pages/viewpage.action?spaceKey=medicine&title=PPI+-+Patient+and+Public+Involvement
Follow on Twitter
- Centre for Sport, Exercise and Osteoarthritis Research Versus Arthritis: @CentreforSEOA
- Centre of Evidence Based Dermatology: @CebdNottm
- Nottingham Biomedical Research Centre Hearing theme: @hearingnihr
- Nottingham Biomedical Research Centre Musculoskeletal theme: @NottmBRCMSK
- Nottingham Clinical Trials Unit: @nottingham_CTU
- Pain Centre Versus Arthritis: @UoNPainCentre
By Dr Adele Horobin with Dr Carron Layfield, Dr Joanne Stocks and Ms Helen Taylor of the School of Medicine patient and public involvement working group
No comments yet, fill out a comment to be the first



Leave a Reply