September 27, 2017, by Dr. Meghan Gray
Levitation in the lab

A guest post by Senior Research Fellow Dr. Richard Hill, whose research recently graced the cover of the September 15 volume of the prestigious journal Physical Review Letters.
I have two superconducting magnets in my lab generating strong magnetic fields that can levitate water, wax, alcoholic drinks, flies, supercarrots, you know, that kind of thing…
You don’t?
What that peculiar list of things has in common is that they’re all diamagnetic. They’re all repelled by magnetic fields, unlike the more commonly known ferromagnetic materials (iron, steel, nickel, novelty fridge magnets), which are strongly attracted. In our magnets, this repulsion counteracts the force of gravity all throughout the diamagnetic object, at the molecular level, so that it ‘feels’ weightless, as an astronaut does in orbit.
This allows us to do experiments in weightlessness that would ordinarily require a spacecraft, or a parabolic ‘vomit comet’ flight.

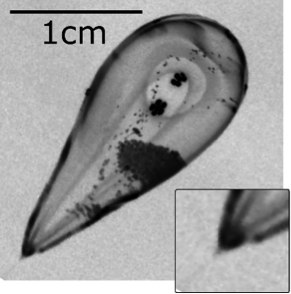
Levitated droplets of water are spherical, just as they are in space. This photograph shows a globe of water, several centimetres in diameter, suspended in mid-air by the strong magnetic field produced by one of our magnets.

We, graduate student Liam Liao and I, levitated liquid droplets to study what happens to them when we spin them. A droplet spun fast enough distorts into a dumbbell shape. Spinning it faster breaks it in the middle into two baby droplets.

In addition, we applied up to 7000 volts to the droplets, electrically charging them. The individual electric charges repel each other but are trapped on the surface of the droplet. When the amount of charge exceeds a critical amount, predicted by the eminent scientist Lord Rayleigh in 1882, the repulsion between them becomes strong enough to burst the droplet in a ‘Coulomb explosion’. The liquid streams out from the surface as needle-like jets.
This process lies at the heart of many important technologies, from crop spraying to electrospray mass spectrometry.
In our experiments, we found that fast rotating and highly charged droplets can break apart either via the dumbbell, the jet or an unusual hybrid of both.

We used our novel method of breaking—fissioning—charged droplets by spinning them to show that these droplets do indeed break up at exactly the amount of charge given by Lord Rayleigh’s prediction, a notoriously difficult thing to test experimentally.
There is a long history, going back to the 19th century, of using droplets as mathematical models of other objects, from spinning planets and stars, to atomic nuclei. Magnetic levitation turns out to be an ideal way to study the real counterparts of these models, in the laboratory!
No comments yet, fill out a comment to be the first

Leave a Reply