
October 29, 2005, by Teaching at Nottingham
Practical molecular biology for visually impaired students
This illustrated report shows the simple adjustments that allow Emily (who has cone-rod dystrophy giving her very little detailed vision but some colour perception) to carry out her own lab work. Each Visually Impaired (VI) student has specific needs but adjustments here may be of use to them.
This was originally written in 24 point Bold Arial by Emily (her preferred font for reading and WP).
Picking colonies, labelling plates and pipetting microlitre volumes
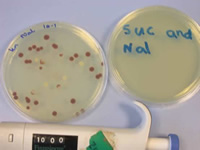
The plate on the left shows individual colonies of 0.4cm in diameter which are easy to pick off especially since they are pigmented colonies.
The plate to the right shows clear labelling that I did with a thick marker pen, which is easy to read.
The pipette at the bottom has a clear white on black number display that I can read (& so accurately adjust) without a magnifier. Gilson pipettes are black on white numbered but can be read with a hand magnifying glass.
Labelling small Eppendorf tubes

This picture shows the labelling of Eppendorfs with white tape and a thick pen. By doing this I can easily find the right Eppendorf.
Aseptically Pipetting broth
 Here I am pipetting up some broth, making sure my tip is at the bottom of the bottle so that I know I have taken up the correct amount of liquid. 40ml in 100ml Duran is ideal for this as it doesn’t go over the top of the tip. Thus the broth is not contaminated.
Here I am pipetting up some broth, making sure my tip is at the bottom of the bottle so that I know I have taken up the correct amount of liquid. 40ml in 100ml Duran is ideal for this as it doesn’t go over the top of the tip. Thus the broth is not contaminated.
Spreading plates

Here I am using a spreader on an agar plate, close to a Bunsen flame, after delivering a sample into the middle of the plate from a pipette.
I can do this aseptically without getting too close to the plate.
The blue disposable spreader is easy to see unlike a glass one.
Line marked cylinders for measuring volumes

In the process of making agar I measured 100mls of water into a measuring cylinder marked with a thick pen at that level and then poured it into a bottle. In even lighting (no glare no shadows) I can see the meniscus.
Pouring plates at a Bunsen


I can pour agar plates well when the agar is dark coloured for a light bench (as in the picture) or light agar with a dark bench this creates good contrast.
I can work near a Bunsen safely and aseptically and light it myself (Modern Bunsen with lever and pilot light is ideal as it has a steady flame)
Showing up E.coli on chromogenic agar
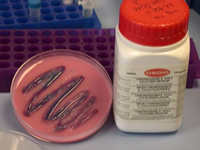
This shows Chromogenic agar (which contains dyes) with E.coli grown on it. The bacteria (commonly used in practicals) show up well because they appear dark on this agar. I can then see where to take a sample from. This is much easier than normal agar which is the same colour as the E.coli. My DSA (Disabled Students Allowance) can be used to buy chromogenic agar and pre-sterilised blue loops (see below) these are more visible for streaking plates than wire loops.


I can see microscopic images of bacteria easily on a video screen hooked up to a microscope. If squares are pre-marked on slides then I can pipette on microlitres of bacteria, cover them with a glass coverslip and put them on the microscope.
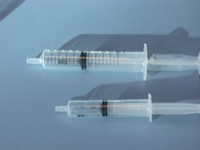
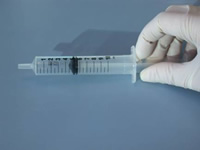
Notches cut into the barrel of syringes to correspond to set volumes allows any VI student to measure liquids (N. Brown RNIB New College Worcester)
Setting up complex PCR reactions using a large protocol list


Emily setting up PCRs using large print protocol and pipetting tiny volumes accurately into tiny but colour-coded tubes
Small tubes are easily handled and mixed


Especially if the tops are labelled with coloured tape to stand out against the ice

Emily needs no special assistance to work in lab practical classes, a regular lab partner is all she requires.
For final year project work, she is well able to work alone on techniques that she has been shown and to produce research level results.
Emily Clarke
School of Biology
Liz Sockett
Professor, Faculty of Medicine & Health Sciences
School of Biology
No comments yet, fill out a comment to be the first

Leave a Reply