
February 24, 2020, by Rob Ounsworth
Taming microbes at the University’s Biodiscovery Institute
On 12 May the University is officially opening its £100 million Biodiscovery Institute.
In the first of a series of blogs, its Director Chris Denning wrote on Valentine’s Day about how the Institute’s expertise in regenerative medicine will help cure broken hearts though its world-leading stem cell research.
Today we’re highlighting another area of cross-disciplinary expertise that will be showcased to colleagues and visitors at the Institute’s launch.
Taming microbes will help our researchers meet global challenges such as antimicrobial resistance (AMR).
Giving bugs the slip
In its previous guise as the Centre for Biomolecular Sciences, the Institute brought together chemical engineers, microbiologists and biomedical experts, who took the discovery of a bacteria-resistant material, through to a product now available for use in the NHS that is capable of preventing catheter associated urinary tract infections – the most common cause of healthcare-acquired infections.
Morgan Alexander, Professor of Biomedical Surfaces, says: “This has gone all the way from a discovery of a new class of materials to clinical trials, and that’s a massive achievement. The CE mark is for urinary catheters only, but we have had a lot of interest from companies who manufacture other medical devices. This is an exciting time.”
The high-resolution image above was captured by an electron microscope and shows minerals formed inside a standard silicone urinary tract catheter through the action of urease producing bacteria. These ultimately block the catheter and are associated with catheter-associated urinary tract infections.
Elsewhere, guests on 12 May will discover other ways in which bio researchers are working towards taming microbes:
The power of polymers
Paul Williams, Professor of Molecular Microbiology, helped develop the new group of bacteria-resistant polymers used in the catheters. Now he is deepening understanding of how bacteria sense and adapt to surfaces, so other life-saving medical implants and devices can be made resistant to the formation of biofilms, reducing infections and the use antibiotics.
Defeating antimicrobial resistance
Dr Kim Hardie’s research is helping to combat the global threat of antimicrobial resistance – by 2050 it is predicted that more people will die from infections than from cancer. She says: “Our groups are using multidisciplinary, state-of-the-art approaches to visualise the effectiveness of antimicrobials as they kill bacteria and design more potent drugs which can combat AMR.”
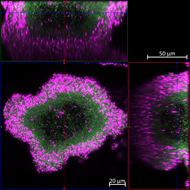
Dr Hardie adds: “This image shows confocal microscopy of a microcolony of bacterium Pseudomonas aeruginosa showing live (green flourescent protein producing) cells at the centre surrounded by pink cells killed as the antibiotic ciprofloxicin penetrates through.”
Silencing harmful bacteria
Professor Miguel Cámara, of the National Biofilms Innovation Centre, is working towards silencing communication between microbes and stop them forming biofilms – the slime microbial cities that cause infectious diseases and resist treatment. He says: “Our researchers are studying ways to interfere with this communication, which will weaken biofilms, stop them causing damage to humans and make them more sensitive to antibiotics.”
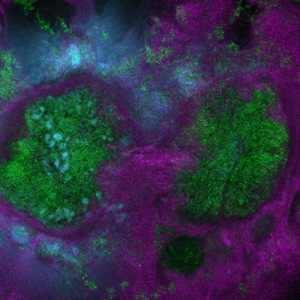
This image shows polymicrobial biofilm of Staphylococcus aureus, Pseudomonas aeruginosa and Candida albicans.
Engineering friendly microbes
Dr Stephan Heeb is harnessing the power of bacteria themselves in the fight against infections. He is exploring how friendly microbes can be engineered and programmed to explore the body and other environments, detecting harmful microorganisms far earlier and leading to swifter, more targeted and effective action against infection.
Black Death still a threat
Black Death is not a disease of the past and unfortunately plague is still with us. Thousands of cases are confirmed each year and it been classified as a ‘re-emerging’ pathogen. Dr Steve Atkinson of the Plague Research Group is studying the genetics behind how plague causes disease and how it survives in the environment. Understanding more about this devastating pathogen will help control future outbreaks.
Professor Chris Denning says: “Here at the Biodiscovery Institute we recognise that complex global challenges such as antimicrobial resistance requires imaginative, truly cross-disciplinary approaches. The work outlined here in ‘taming microbes’ illustrates how our world-leading researchers tackle such problems from many angles, while focused by a shared sense of purposeful collaboration.
“I look forward to sharing further exciting projects with you as we look forward to officially opening our doors on 12 May.”
No comments yet, fill out a comment to be the first

Leave a Reply