December 15, 2015, by Peter Cartwright
A Headache for Nurofen?
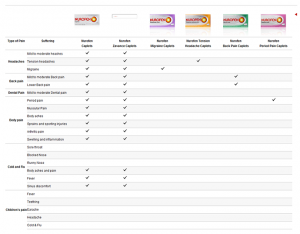
The Federal Court in Australia has found that Reckitt Benckiser (Australia) breached Australian consumer law by marketing Nurofen Specific Pain products as formulated to treat a specific type of pain, when the products were identical in terms of their active ingredient. The products in question were: Nurofen Back Pain, Nurofen Period Pain, Nurofen Migraine Pain and Nurofen Tension Headache. The products all contained the same active ingredient, (ibuprofen lysine 342mg). None was more effective at treating the specified pain than the other Nurofen Specific Pain products.
The misleading nature of the marketing was particularly demonstrated by the details on the Nurofen website in September 2013 (since amended but see below).
Source https://www.accc.gov.au/media-release/accc-targets-alleged-false-and-misleading-nurofen-claims
According to the company, no changes will be made to the packaging in the UK. I had a look at the UK website yesterday (14-12-15): http://www.nurofen.co.uk/our-products?adult-pain It described three products as “symptom specific” and two (Nurofen Migraine Pain and Nurofen Express Period Pain) as providing “targeted relief”.
I have been pondering how UK consumer law might address this. The Consumer Protection from Unfair Trading Regulations 2008 (CPUTRs) contain a number of provisions that might be relevant. First, the CPUTRs prohibit misleading actions. Regulation 5(2) states that a commercial practice is a misleading action:
“(a) if it contains false information and is therefore untruthful in relation to any of the matters in paragraph (4) or if it or its overall presentation in any way deceives or is likely to deceive the average consumer in relation to any of the matters in that paragraph, even if the information is factually correct; and
(b) it causes or is likely to cause the average consumer to take a transactional decision he would not have taken otherwise.”
Paragraph (4) refers to a rage of matters. Among them are “the main characteristics of the products” which includes “the benefits of the product” and the “fitness for purpose of the product”. So, might we argue that by describing a product as targeting specific pain when it is no more effective at tackling that pain than the other products, the company has engaged in a misleading action? There are two elements to consider.
First, is its overall presentation likely to deceive the average consumer in relation to any of the matters in that paragraph? If an average consumer is likely to believe that a Nurofen Migraine Pain product was more likely to be successful in tackling migraine pain than a Nurofen Back Pain product then the answer to this seems to be yes. An average consumer for the purposes of the CPUTRs is one who is “reasonably well-informed, reasonably observant and circumspect”. It is certainly arguable that such a consumer would assume the targeted product to be more fit for a particular purpose than the others. Furthermore, the Regulations state that when a product is aimed at a particular group of consumers, the average consumer is the average member of that group. It could be argued that the product is aimed at the average consumer who suffers from that particular ailment and that such consumers will be particularly eager to find a product that deals with their condition. As Suzanne Moore commented in her piece in today’s Guardian: “When ill or in pain, we will pay for any promise of a magic bullet”.
http://www.theguardian.com/commentisfree/2015/dec/14/nurofen-targeted-painkillers-reckitt-benckiser?CMP=twt_gu
Second is the misleading description likely to cause the average consumer to take a transactional decision he would not have taken otherwise? As one example of a transational decision is buying a product, it seems likely that the description of the product as “symptom specific” and providing “targeted relief” is likely to lead the average consumer with a migraine to purchase the product. That is, presumably, what the company intends. Why else would it package the products as it did or present the information on the website as it did? According to a statement from the company, the specific-pain range’s purpose was “to help consumers navigate their pain relief options, particularly within the grocery environment where there is no healthcare professional to assist decision making”. This does not appear particularly convincing. The Medicines and Healthcare Products Regulatory Agency has stated that it has not received complaints about Nurofen in the UK, though whether that will remain the case following the amount of media coverage the Australian case has received remains to be seen.
The front page of the Independent reports today (15-12-15) that the Advertising Standards Authority is examining complaints that television advertisements for Nurofen Express are misleading because they (a) imply that the product directly targets muscles in the head, and (b) give faster relief than standard paracetamol or ibuprofen.
It should be noted that there is no suggestion that the product is inappropriate for the particular purpose. What the company appears to have done is created the impression that one product is more appropriate for a particular condition than the others; and that, the Australian Federal Court found, was not true.
The Australian case has produced considerable comment already. Writing in the Conversation, Michael Vagg notes that the Australian Therapeutic Goods Administration first ordered the claims to be withdrawn in 2011 and speculates that the company knew that it would have to change its marketing if the ACCC instituted proceedings. However, he muses, it may not have wanted to jump before it were pushed (proceedings were first instituted by the ACCC in March 2015). http://theconversation.com/can-the-accc-target-the-source-of-misleading-labelling-52303
Vagg is (understandably) unsympathetic to the company. By contrast, many of the comments posted by way of discussion have been extraordinarily unsympathetic to consumers who have fallen foul of the company’s actions. Words such as “stupid” “idiots” and “ignorant” have been thrown around. It remains to be seen whether the UK authorities are so unsympathetic and also, whether the negative publicity the company is receiving has significant impact on its company’s sales.
No comments yet, fill out a comment to be the first

Leave a Reply