August 23, 2018, by Brigitte Nerlich
Phage and fiction
We have known about bacteriophages for over a century. I myself became vaguely aware of them around 2004 when I started to be interested in bacteria and antimicrobial resistance and later on when my mother had Clostridium difficile, a health-care associated infection related to antibiotic use. However, I never actually looked more closely at phages until Carmen McLeod, my colleague at the Synthetic Biology Research Centre, Nottingham, drew my attention to them again more recently.
I knew, of course, that some of my colleagues at the SBRC use phages when trying to find therapies for Clostridium difficile. Here is Micky explaining her work with ‘Dave the phage’, for example. And our 2018 iGEM team also uses phages. Their project is entitled “Engineering bacteriophage to silence toxin expression in Clostridium difficile”!!
But what are phages and why are they so intriguing and important in science and society? In this post I’ll weave between fact and fiction, as I found that phages are not only fascinating in term of science and potential therapies, but also in terms of aesthetics and fictional potential. And, of course, when we think about or use phages, fact and fiction might begin to interact.
Phages, science and history
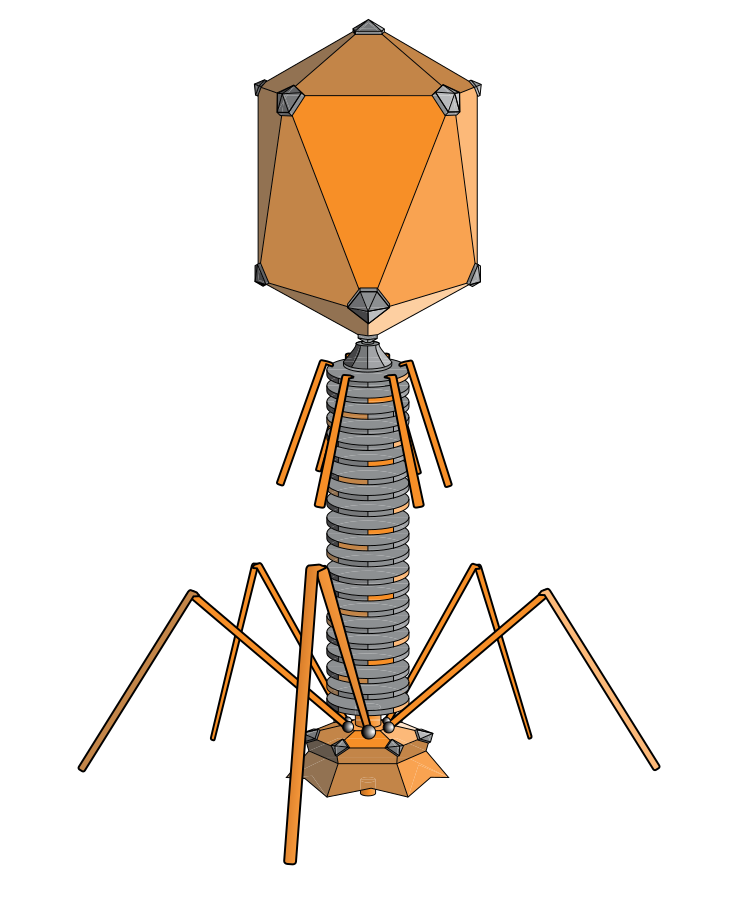
An article in The Lancet provides a good definition of what bacteriophages are (for more info read this book by Carl Zimmer): “A phage is a tiny virus that latches its fibres onto a bacterium* and impregnates it with DNA, hijacking the machinery of the cell and forcing it to produce countless more phages which eventually burst out, destroying the bacterium.” Phages ‘eat’ bacteria…They are also said to kill, annihilate, slay, wage war against bacteria…
As bacteria are developing resistance to antibiotic drugs, phages are explored as an alternative to antibiotics. (I won’t go into the war, weapon and armamentarium metaphors often used in this context, but that deserves some attention). “Therapeutic phages are naturally occurring viruses that can be selected to kill only specific bacterial species or strains while leaving other, helpful bacteria and mammalian cells unharmed. Thus, unlike broad-spectrum antibiotics, they spare the commensal microbiota (e.g., in the gut)—a characteristic particularly important for malnourished and immunocompromised individuals.” This is also important in the context of Cdiff…
Now a bit of history: “Phages were discovered in 1915 by British microbiologist Frederick Twort, and, independently in 1917, by French-Canadian microbiologist Felix d’Hérelle. Twort did not pursue his discovery, whereas d’Hérelle systematically investigated the nature of bacteriophages and explored their ability to function as therapeutic agents.” Basically, things happened between 1915 and 1919….(and possibly even before).
Things happened especially in Russia because, as the Lancet article points out, “D’Herelle struck up a partnership with a Georgian scientist named George Eliava; the pair later worked together in Tbilisi, where the Eliava Institute was founded in 1923. Early results were promising and western companies soon began to market phage therapies for typhoid and urinary-tract infections. Then, with Alexander Fleming’s discovery of penicillin in 1928, interest rapidly died out”, but not in Georgia, where phage therapy continued to flourish and is having a revival just now.
In fact, phage therapy seems to be having a bit of a global renaissance in the context of the rise in antimicrobial resistance. Whether it will really work is another question…
Phages, art and fiction
If you clicked through some of the articles mentioned above you’ll have seen some wonderful pictures of phages. Aren’t they fantastic? Who would have thought these viruses were natural and not made up by some sci-fi enthusiast? The following is probably a typical reaction upon first seeing an image of a bacteriophage: “What was this? […] that bacteriophage was like something out of Deep Space Nine or The Navigator. This ‘bacteriophage’ was clearly some comedian’s idea of a microbial-jackalope: a cross between a spacecraft and a spider. It was scary looking!”
Scary looking, that’s what bacteriophages are. Here are some arty renditions reproduced in the Smithsonian Magazine and here are some more in the Science Photo Gallery…. (and here). All this would lend itself to some nice visual analysis!
Another avenue for future research would be the depiction of phages in fiction, especially science fiction. One of the books that comes up in this context is by Mark Tamplin, simply called Phage. There is also a Star Trek episode it seems, and phages moved into video games too. There are even lovely Lego models of fictional phages.
But phages are not only scary looking; they are also just scary or sometimes portrayed as such, as in this YouTube video entitled “The Deadliest Being on Planet Earth – The Bacteriophage”, which is worth some analysis too!
Phages, science and fiction
One of the most well-known fictional treatments of phage therapy can be found in the 1925 novel Arrowsmith by Sinclair Lewis, which has been nicely discussed in this article by Ilana Löwy.
When rummaging around on the web, I read a blogpost that revealed a very intriguing interaction between science and science fiction.
In 1957 Hal Clement wrote a science fiction novel entitled Cycle of Fire set on the planet Abyormen “which orbits a dwarf star, which in turn circles a blue giant. The planet’s usual orbit creates two sixty-five-year-long seasons; the ‘hot season’ and the ‘cold season’. Each season is dominated by a different intelligent species.”
As a scifi website explains: “One of the most interesting aspects concerning the lifeforms of Abyormen is the way they reproduce. This happens only during the passage from one season to another. Both Abyormenite species reproduce in a way analogous to bacteriophage viruses on Earth: by laying reproductive spores inside the cells of other creatures. When the ‘hot season’ comes to end, all ‘hot Abyormenites’ die, and the new generation of ‘cold Abyomenites’ start emerging from their bodies, in which their ancestors had left their spores 65 Earth years before. During their life, these ‘cold beings’, in turn, carry the dormant spores of their ‘hot’ counterparts inside each cell of their body. And when they themselves finally die at the end of the ‘cold season’ it is the ‘hot Abyormenites’ who emerge, completing the cycle. As a result, both races are so deeply linked to each other in this symbiosis that they depend upon the death of each other to reproduce.”
Interestingly, this novel was inspired by a scientific article about viruses that infect bacteria, that is, bacteriophages. The article was by André Lwoff and had the title “The life cycle of a virus”. It was published in March 1954 in Scientific American. Lwoff received the Nobel Prize in Physiology or Medicine in 1965 for his research on bacteriophages!
As Hassler points out in his book about Hal Clement: “Lwoff describes the way certain viruses are capable of remaining dormant inside the totally alien life form of certain bacteria for several generations before they reappear as distinct virus life, and he emphasizes the cycles of life and death coupled here with reproduction the protection of generation. Although much of Clement’s fiction avoids such sublime and poetic images, Cycle of Fire is meant to demonstrate, on this most elemental level of life and death (not microscopic in the novel, of course), the symbiotic inter-dependence of what seem to be very alien life forms.” (p. 33)
Symbiosis may, of course, not only be important for alien life forms, but for us too, as microbiome and phage experts are discovering. As Carl Zimmer wrote in 2013: “Meet Your New Symbionts: Trillions of Viruses“.
Phages, synbio and origin stories
So, work on phages has been going on a long time and is even related to the beginnings of molecular biology. There are still lots of mysteries surrounding these life forms, if indeed they are life forms – some scientists even use the metaphor of ‘viral dark matter’. But what has all this to do with synthetic biology? I ask this question briefly, as I am supposed to be part of the SBRC after all. A real answer to this question, would, yet again, be quite a long one.
The short answer is: phages are linked to one, and I stress, one, of the origin stories told about synthetic biology and also about the discovery of CRISPR (see here).
This was brought home to me when reading a review of Sophia Roosth’s book Synthetic: How life got made, based on lab ethnography. The review is by Philip Ball and Ball writes:
“Roosth’s epiphany seems to have occurred after observing the way [Drew] Endy responded to his early work on computational modeling of the bacteriophage T7, a virus that infects bacteria. His model took into account all that was then known about the virus’s genetics to try to predict so seemingly simple and fundamental a property as its growth rate. But the model failed dismally.
Most researchers would have concluded that either they had got the modeling wrong or there was still some crucial information about the virus that was lacking—in any event, that the theory had shortcomings. But Endy’s view was different. The problem, he decided, was the virus. It was too damned complicated. The modeling would work fine if the virus could be changed to fit it. ‘Because the model did not work,’ Roosth writes, ‘instead of scrapping the model, Endy decided to modify the virus.’
This, she says, amounts to a conclusion that ‘Nature with a capital N’ isn’t so interesting in itself. What matters is the ability to refashion it ‘as it should be.’”
Hence, synthetic biology. You can find more info on this origin story in the article by Rebecca Wilbanks for Aeon Magazine entitled: “If we made life in a lab, would we understand it differently?” She ends the article by saying: “”What new forms life will take, only time will tell.” And what contribution phages will make to life and death, only time will tell.
*I have replaced bacteria by bacterium; sorry Lancet!
Image: Wikimedia Commons
No comments yet, fill out a comment to be the first

Leave a Reply